Let's Get Direct: How working directly with patients optimizes delivery of complete RWD
Real-world data (RWD) and real-world evidence (RWE) continue to gain traction within clinical research, supporting all stages of the therapeutic development process and influencing health care decisions. The US Food and Drug Administration (FDA) has issued several recent guidance documents that address quality and data standards for the use of RWD and RWE in regulatory decision-making. This is an exciting time, and PicnicHealth was delighted to act as a Silver Sponsor for the annual Real-world Evidence and Life Sciences Analytics Conference hosted by MarketsandMarkets in Boston this past October.
It was wonderful to convene with industry leaders to discuss the varying perspectives of stakeholders across the ecosystem, innovative strategies for demonstrating value, best practices for leveraging artificial intelligence and machine learning, and many more intriguing topics.
Our very own Kristen Hahn, PhD, MPH - Head of RWE Research, gave a captivating presentation, “Let’s Get Direct: How working directly with patients optimizes delivery of complete RWD” where she:
- Outlined the PicnicHealth patient-centric process for patient recruitment, consent and retention
- Described how we incorporate patient centricity end-to-end to enable data quality and completeness
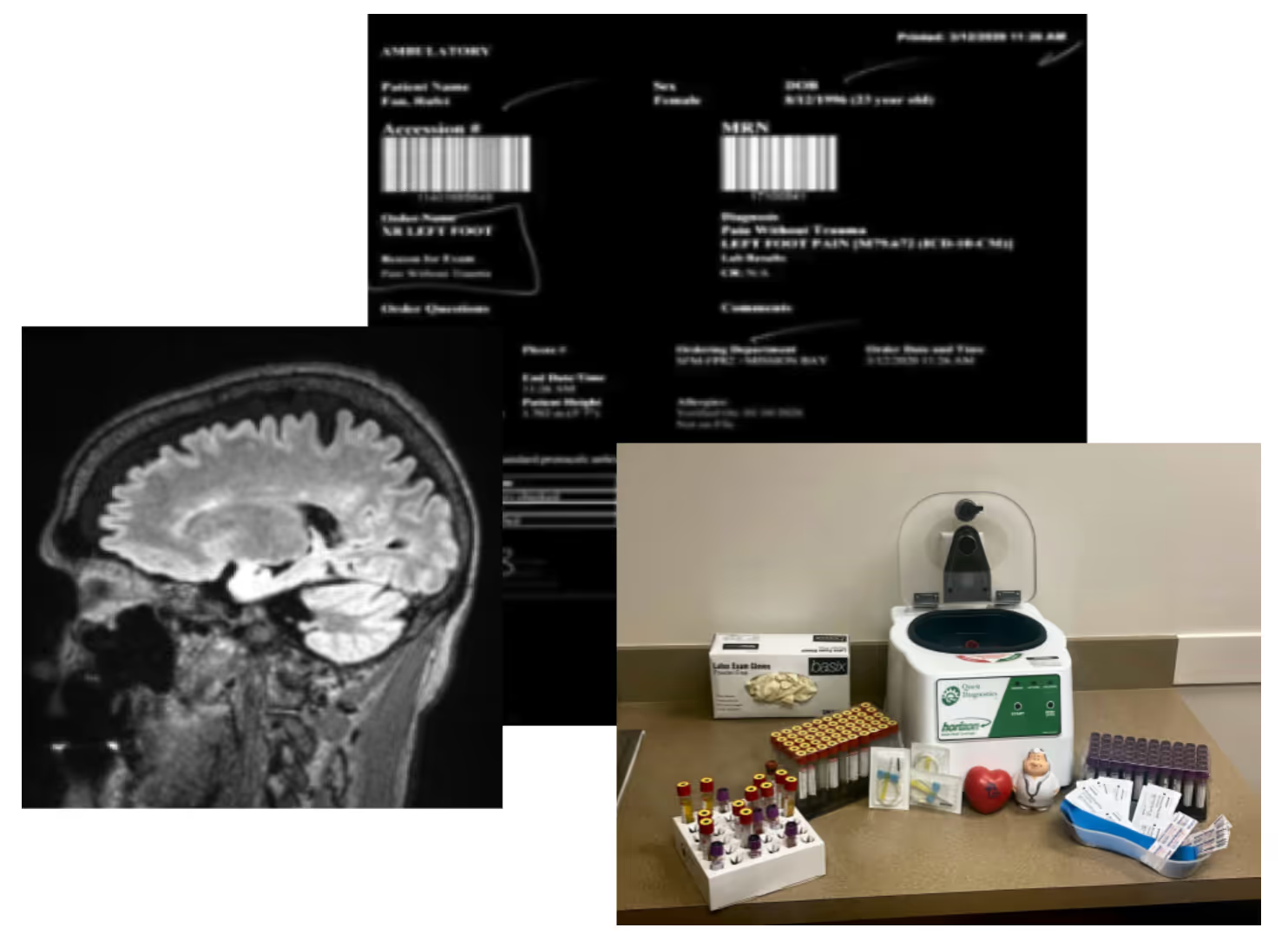
- Shared real examples from our Multiple Sclerosis research cohorts that use direct-to-patient approaches
View her full presentation below.
Interested in learning more about our research cohorts chronic and complex diseases in several neurological and hematological indications and select rare diseases? Contact a PicnicHealth expert and visit our Research Platform website.

























































































.avif)
