View Transcript
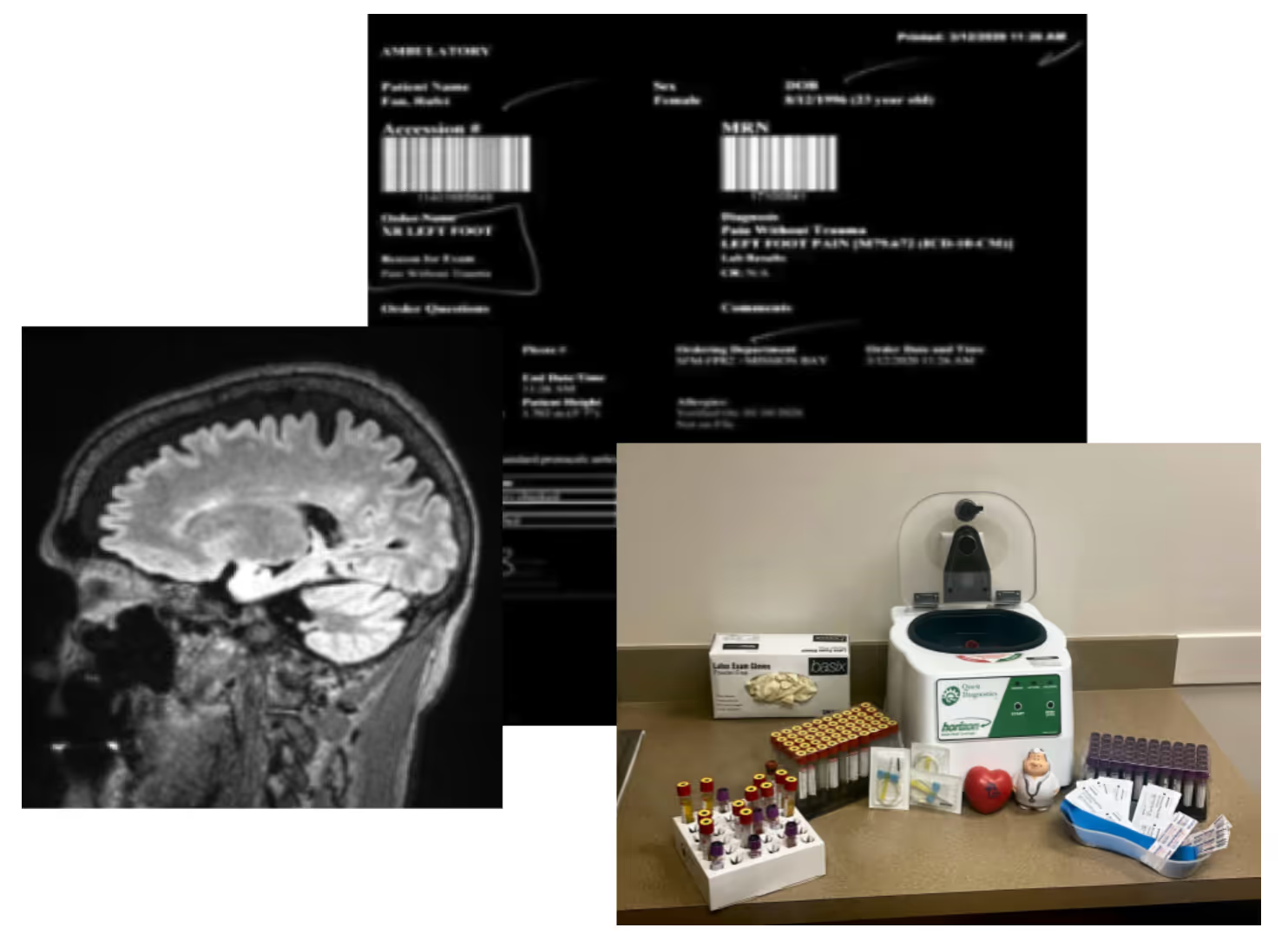
Long-term follow-up (LTFU) studies are often a complicated and costly requirement for life sciences companies, but they can be greatly simplified with the advent of AI and other new technologies. With regulators requiring more than ten years of follow-up for certain therapies, there is a critical need to implement these new methods of collecting safety and effectiveness data to reduce burden and costs on sponsors, patients, and sites. In this webinar, panelists discuss what new technology-enabled processes look like, the research challenges they overcome, and considerations to increase patient retention, study efficiency, and data quality.
Key Learnings:
Learn the benefits of:
- Streamlining LTFU studies and decreasing burden on sponsors, patients, and sites
- Engaging patients in multi-year studies to decrease loss to follow-up
- Leveraging technology like AI to ensure high-quality endpoint collection
- Transitioning existing LTFU studies to tech-enabled methods and the benefits of doing so
{{panelists}}