In this issue, we're featuring a perspective from Marisa Co, strategic advisor to PicnicHealth. Marisa has more than 30 years driving financial strategy, data-driven decision-making, and business transformation in global and small healthcare organizations. Below, she makes the case for why the convergence of AI and patient-mediated data isn't an incremental improvement. It's a restructuring.
The future of clinical research is patient-mediated — and it always should have been. For too long, the industry has treated patients as a means to an end: data sources to be extracted from, not collaborators to be engaged. That model is inefficient, inequitable, and increasingly obsolete. When patients are given control over their own health data and invited into the research process as active participants, the quality of evidence improves, retention increases, and the findings actually reflect how people live. This is not a speculative vision. It is already happening. And AI is about to accelerate it beyond anything the industry is prepared for.
My passion has always been using technology to make clinical research more efficient, humane, and accessible. In 2009, I helped build one of the first platforms enabling fully remote trial participation — electronic informed consent, web-based data collection, and home-based study designs — long before "decentralized trials" entered the mainstream vocabulary. The insight that drove us then holds just as true today: when you respect patients' time and give them intuitive tools, they engage deeply and generate data that is richer and more representative of real life.
From Subjects to Collaborators
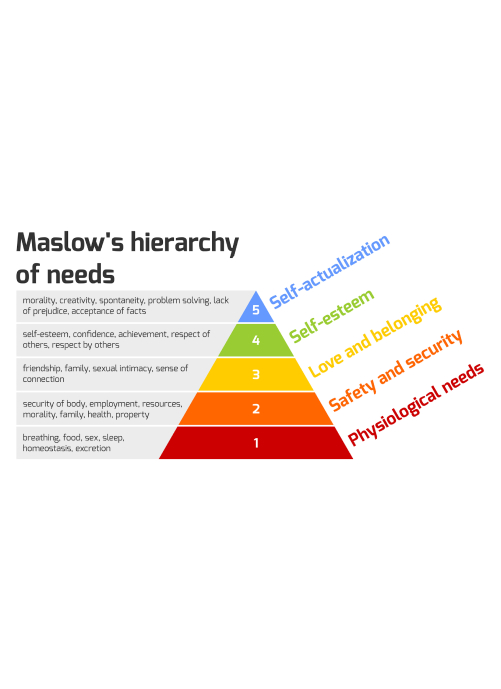
Rather than treating people as passive "subjects," patient-mediated research engages them as collaborators, giving them control over how their data are collected, aggregated, and shared. PicnicHealth extends this philosophy into non-interventional resrach, assembling medical records from all of a patient's sites of care into a unified longitudinal record, pairing comprehensive clinical histories with patient-reported outcomes and patient-generated data to give researchers a far more complete picture of each person's health journey. Direct-to-patient registries can enroll virtually, follow participants over time, and give patients access to their own data for use in their own health journeys.
What makes this moment genuinely historic is the collision of patient-mediated data infrastructure with AI that is advancing faster than the research industry is prepared for. We are not simply automating existing workflows. We are approaching a fundamental restructuring of how evidence is generated.
Where We’re Going — Fast
Here is where I believe we are headed, and sooner than most expect:
The randomized controlled trial will no longer be the default gold standard for many indications.
AI models trained on deep, longitudinal, patient-mediated datasets will generate synthetic control arms so statistically robust that regulators will increasingly accept them in place of placebo groups — sparing thousands of patients from receiving no treatment while accelerating approval timelines by years. The FDA and EMA are already moving in this direction, and within a decade this will be standard practice across multiple disease areas.
AI will make n-of-1 research scalable.
Today, personalized medicine is largely aspirational. Tomorrow, continuous patient-mediated data streams — wearables, patient-reported outcomes, medical records updated in near real time — will feed AI systems capable of modeling treatment response at the individual level. Sponsors will be able to identify which patients are most likely to respond to a therapy before a trial even begins, collapsing the distinction between research and clinical care.
The research visit will become largely obsolete.
AI-powered digital biomarkers derived from smartphone sensors, voice patterns, and passive behavioral data will replace many of the measurements that today require a clinic. Combined with patient-mediated record assembly, a participant's entire disease trajectory will be continuously observable without a single trial site visit, making research invisible, ambient, and perpetual.
Patients will become active co-investigators.
AI interfaces will give individuals plain-language access to their own longitudinal health data, enabling them to spot patterns, ask questions, and flag insights that researchers may have missed. This is not a distant vision. It is the logical endpoint of patient-mediated models, and it will produce a category of real-world evidence that has never existed before: evidence that patients themselves helped discover.
The Patient Was Always the Point
The research industry has spent decades optimizing a model built around institutions — sites, investigators, sponsors — with patients as inputs. What patient-mediated infrastructure makes possible, and what AI now makes inevitable, is a complete inversion of that model. The patient becomes the constant. The data becomes continuous. And the evidence that results is more complete, more representative, and more actionable than anything the old model could produce.
This is not a threat to rigorous science. It is rigorous science, finally built around the people it is meant to serve. The organizations that recognize this shift early and build for it will define the next era of drug development. The ones that don't will spend the next decade catching up.
The patient was always the point for PicnicHealth. We’re already operationalizing this vision, assembling deep, longitudinal patient records, pairing them with patient-reported outcomes, and powering non-interventional studies that put patients at the center. Contact us for more info.