Recognizing the need for accelerating medical product development and modernizing the drug approval process, the 21st Century CURES Act was enacted in 2016 and augmented the ability for the FDA to incorporate real world data and patient perspectives into the drug approval process. While randomized clinical trials are still the gold standard, observational data is increasingly utilized to supplement clinical trial design, as synthetic controls in single arm trials and for label expansion.
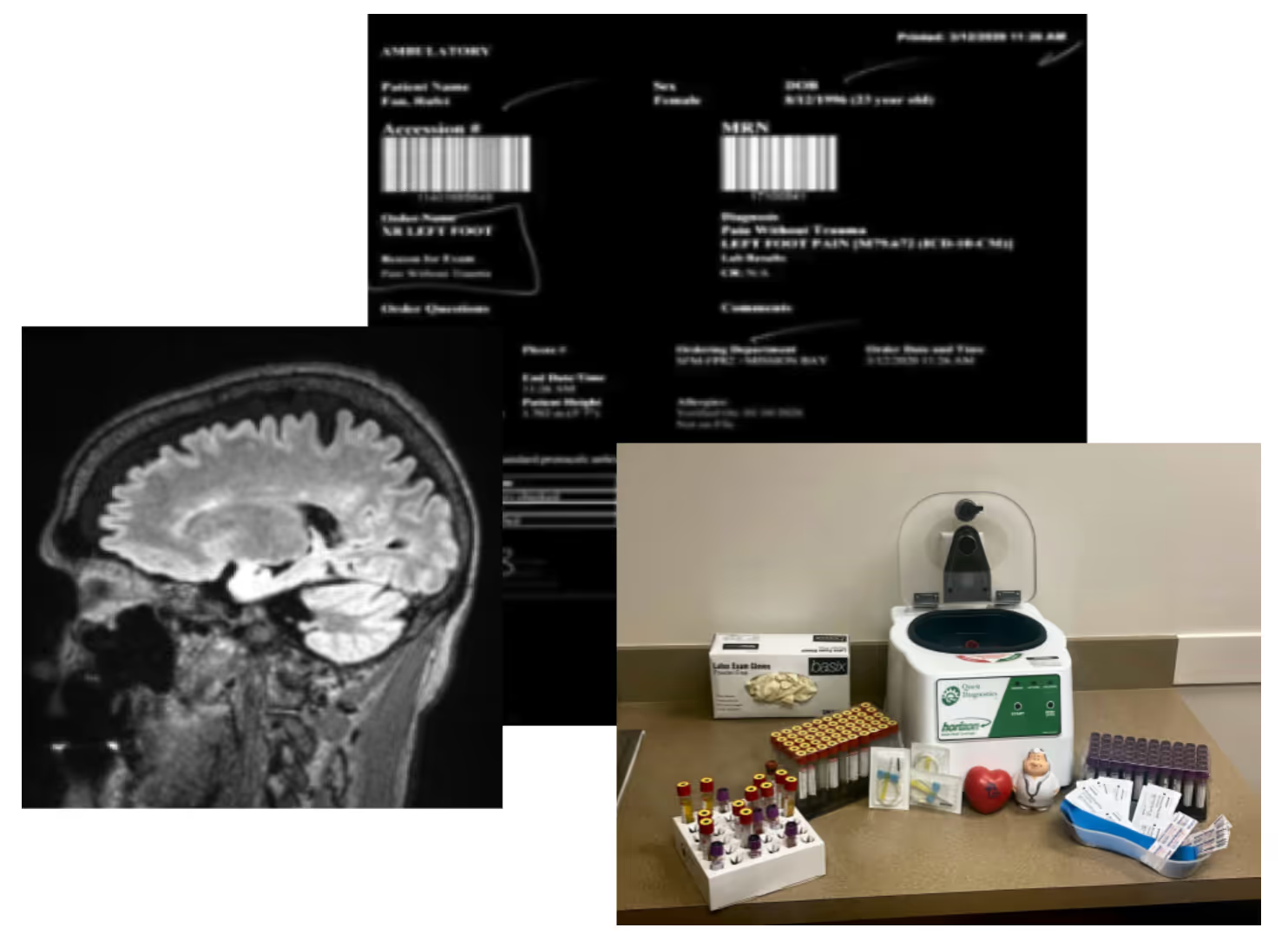
Currently, most existing real world databases are either incomplete (i.e. claims data, system-based EHR data) or expensive and time-consuming to maintain (i.e. registries). PicnicHealth fills the gap by combining insights from electronic health records, claims data, and patient reported outcomes (PROs) for any patient from any facility within the U.S. By providing disease-specific structuring of narrative text sections of records, PicnicHealth is able to unlock the wealth of data that has historically been inaccessible to researchers. On average, PicnicHealth collects five to seven years of retrospective longitudinal patient data for each patient and can continue to collect prospective data as needed.
Learn more in our whitepaper